Key Outcomes:
- Novel cell and gene approaches advanced, with Quell’s CAR‑Tregs entering P1/2 in RA/SSc and YolTech’s in vivo base‑editing therapy for AATD cleared for a global P2/3, signaling growing regulatory comfort with next‑gen modalities
- In metabolic and dermatology, AbbVie’s non‑incretin amylin analog ABBV‑295 showed meaningful P1 weight loss
- The immunology/rheum space saw expansion of targeted orals and rare‑disease pipelines, with SOTYKTU approved in PsA, rilzabrutinib gaining ODD for IgG4‑RD in Japan, orexin agonist oveporexton filed in Japan for NT1, and a bispecific (HBM7575/SKB575) entering the clinic in AD
- Oncology and heme updates were mixed: XPOVIO secured broader MM reimbursement in South Korea, while Immutep’s efti combo failed futility in 1L NSCLC, prompting trial discontinuation but extending cash runway, while Medicus’ SkinJect microneedle doxorubicin patch achieved strong P2 clearance rates in nodular BCC, supporting further development and partnering plans
- On the BD/platform side, Samsung Bioepis partnered with G2GBIO on a long‑acting semaglutide microsphere platform, Redx gained FTD for GI‑restricted ROCK inhibitor RXC008 in fibrostenotic Crohn’s, and Oryzon extended long‑dated IP for iadademstat combination regimens, all enhancing future partnering and lifecycle options

March 03, 2026
Quell Therapeutics Starts P1/2 CHILL Study of QEL-005
Quell Therapeutics has started a P1/2 CHILL trial to evaluate its investigational QEL-005 (CAR-Treg cell therapy) in adults with refractory rheumatoid arthritis and systemic sclerosis following approval of its clinical trial application in the UK. The multinational study will enroll up to 16 participants across the UK, Germany, and Spain, with initial clinical data anticipated in Q1’27. QEL-005 is engineered to be activated in inflamed tissues and lymphoid structures, where it modulates key immune cells, including T cells, B cells, and macrophages, through a regulatory rather than cytotoxic mechanism, aiming to ‘CHILL, not KILL’ inflammation. Preclinical data presented at the American College of Rheumatology meeting showed broad immunomodulatory activity across immune cell types, and earlier clinical evidence from the company’s P1/2 LIBERATE study for liver transplant patients supports the safety and persistence of Quell’s phenotype-locked CAR-Treg cells.
Source: Quell Therapeutics PR Mar 03, 2026
March 05, 2026
Medicus Pharma Releases Positive Top-line Data of P2 SKNJCT-003 Study
Medicus Pharma reported positive topline data from P2 SKNJCT-003 study evaluating doxorubicin microneedle array (D-MNA) therapy (SkinJect) for the non-invasive treatment of nodular basal cell carcinoma (BCC). The randomized, double-blind, placebo-controlled study enrolled 90 patients vs. PBO and 100 μg and 200 μg doses of the microneedle treatment.
The 200 μg cohort showed the strongest efficacy at day 57, achieving 73% clinical clearance and 40% histological clearance, outperforming both the PBO and 100 μg groups. Response rates improved between day 29 and day 57, indicating sustained biological activity of the treatment over time.
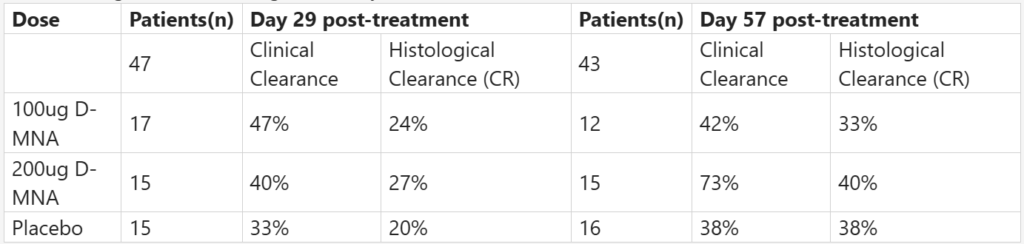
The company considers the results “decision-grade” evidence of clinical activity and plans to complete the full clinical study report (CSR) in Q2 2026 to support a potential end-of-P2 meeting with the FDA. The data may also help advance partnering discussions as the company aims to license or collaborate with larger pharmaceutical partners for late-stage development and commercialization.
Source: Global NewsWire PR Mar 05, 2026
March 09, 2026
Positive Topline Data from P1 GUC17-01 Study
AbbVie released positive topline data for ABBV-295 (SC) from MAD part of its P1 GUC17-01 study, evaluating safety, tolerability, PK, and PD in adults with a mean BMI ≤30 kg/m2, demonstrating clinically meaningful, dose-dependent weight loss over 12–13 wks. with both weekly and extended dosing schedules. The therapy was generally well tolerated, with no SAEs reported and mostly mild gastrointestinal side effects observed early in treatment. These results support further clinical development of ABBV-295 as a potential non-incretin obesity treatment targeting amylin and calcitonin receptors.
Safety Results (Percent Change from Baseline in Body Weight at wk 12 and wk 13)
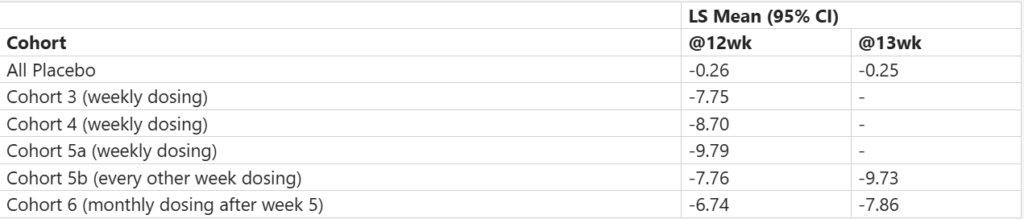
Results from the SAD portion and additional MAD cohorts were reported previously, with full study data expected to be presented at a future scientific conference.
Source: PR Newswire PR Mar 09, 2026
March 13, 2026
Immutep Discontinues P3 TACTI-004 1L NSCLC Study Due to Futility
Immutep has decided to discontinue its P3 TACTI-004 study of eftilagimod alfa in 1L NSCLC, following a recommendation from the Independent Data Monitoring Committee after a planned interim futility analysis. Patient enrollment will stop, and the study will be wound down with appropriate follow-up and site closure. The company is reviewing the data to determine next steps while remaining focused on advancing its broader pipeline. After discontinuing the TACTI-004 trial, Immutep expects its cash runway to extend beyond the previously projected Q2 2027 timeline.
Source: Global Newswire PR Mar 13, 2026

March 01, 2026
XPOVIO Receives NHIS Reimbursement Approval for Multiple Myeloma in South Korea
Antengene reported that South Korea’s National Health Insurance Service (NHIS) has approved reimbursement for XPOVIO (selinexor) + Vd. The regimen is now covered for adults with multiple myeloma (1L+), effective Mar 1, 2026. This marks the 1st reimbursement of an XPO1 inhibitor in South Korea for MM and the 2nd reimbursed indication for XPOVIO in South Korea (MM & DLBCL). Currently, XPOVIO has received approval for three indications in South Korea, including MM (1L+ & R/R) and DLBCL. Antengene gained development, manufacturing, and commercialization rights from Karyopharm in May’18 for China and Macau.
Source: PR Newswire Mar 01, 2026
March 02, 2026
Japan’s MHLW Grants ODD to Rilzabrutinib
Japan’s MHLW has granted ODD to Rilzabrutinib (oral BTKi) for the treatment of IgG4-related disease (IgG4-RD). The decision was supported by positive data from the P2 study and presented at the EULAR 2025 congress. Rilzabrutinib demonstrated reduced disease flares, improvement in disease markers, and decreased reliance on glucocorticoids over 52 wks, with a safety profile consistent with earlier studies and no new safety concerns. The most common AEs include diarrhea, COVID-19, dizziness, dry mouth, and nausea. Rilzabrutinib is now being further evaluated in the P3 RILIEF study. Rilzabrutinib is approved for immune thrombocytopenia (ITP) in the US, EU, and UAE, is under regulatory review for ITP in Japan, and has received multiple expedited designations worldwide for several rare immune-related conditions, including IgG4-RD, WIHA, and SCF, though these additional indications remain investigational.
Source: Global NewsWire PR Mar 02, 2026
March 04, 2026
Takeda Submits NDA for Oveporexton (TAK-861) to MHLW
Takeda submitted an NDA for Oveporexton (TAK-861) to MHLW for narcolepsy type 1 (NT1) in Japan. The application was based on P3 FirstLight & RadiantLight studies. Oveporexton is an oral selective agonist targeting orexin type 2 receptors, aimed at restoring orexin signaling linked to narcolepsy type 1 (NT1). It has received breakthrough therapy designation for treating excessive daytime sleepiness associated with NT1 from both the U.S. FDA and NMPA (China) and is recognized as a Pioneering (Sakigake) and Orphan Drug Designation for NT1 by MHLW (Japan).
Source: Takeda PR Mar 04, 2026
March 06, 2026
SOTYKTU (deucravacitinib) Approves by U.S. FDA for Active Psoriatic Arthritis (PsA) in adults
The US FDA approved BMS’s SOTYKTU (deucravacitinib) for active psoriatic arthritis (PsA) in adults. The approval was based on P3 POETYK PsA-1 and POETYK PsA-2 studies, which demonstrated significant efficacy and a consistent safety profile for SOTYKTU 6 mg OD.
Efficacy Results at Week 16:
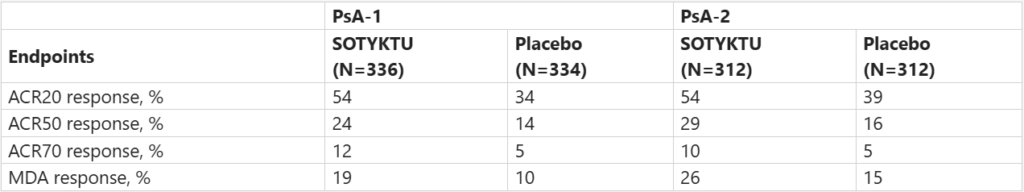
Safety Profile
The overall safety profile in active PsA was consistent with that observed in plaque psoriasis.
Common adverse reactions (≥1%): Upper respiratory infections, increased creatine phosphokinase (CPK), Herpes simplex, Mouth ulcers, Folliculitis, Acne.
Source: BMS PR Mar 06, 2026
March 09, 2026
HBM7575/SKB575 Receives IND Approval from NMPA (China) for Atopic Dermatitis
Harbour BioMed and Kelun-Biotech received IND approval from the NMPA for HBM7575/SKB575, a long-acting BsAb targeting thymic stromal lymphopoietin (TSLP) and an undisclosed target for the treatment of atopic dermatitis. The therapy aims to address unmet needs in moderate-to-severe disease where current treatments often lack durable control. Kelun-Biotech will lead global development and commercialization, while Harbour BioMed will support investment and co-development.
Source: PR Newswire PR Mar 09, 2026
March 10, 2026
US FDA Grants FTD to RXC008 for Fibrostenotic Crohn’s Disease
Redx Pharma received FTD from US FDA for RXC008, a GI-restricted pan-ROCK inhibitor being developed as a potential FIC treatment for fibrostenotic Crohn’s disease. The designation was supported by encouraging preclinical and P1 data. Redx Pharma is working with the U.S. FDA and the STAR Consortium to define the regulatory pathway and plans to initiate a P2 study following positive preclinical and P1 data.
Source: Redx Pharma PR Mar 10, 2026
March 14, 2026
YOLT-202 to Enter Clinic Globally for AATD Patients
YolTech Therapeutics has received IND approval from the U.S. FDA for its investigational in vivo base-editing therapy, YOLT-202, to treat Alpha-1 Antitrypsin Deficiency (AATD). YolTech plans to launch an open-label, single-dose expansion P2/3 clinical trial to evaluate YOLT-202’s efficacy and safety in adult AATD patients. This global trial to be conducted across sites in the U.S. and internationally. Currently, YOLT-202 is undergoing an FIH Early Phase study to assess its safety, tolerability, and preliminary efficacy. Recently, two patients have already completed the dosing, revealing significant dose-dependent increases in AAT levels within one wk. These levels not only exceeded the protective threshold of 11 μM but also reached normal values (>20 μM) in the 45 mg cohort. Additionally, the AAT produced was structurally corrected and functional, with over 95% of the M-AAT showing correction in the 45 mg group.
Source: Businesswire PR Mar 14, 2026

March 11, 2026
Tempest Advances TPST‑2003 toward U.S. IND with New Manufacturing Partnership
Tempest Therapeutics partnered with the Cincinnati Children’s Applied Gene and Cell Therapy Center to support technology transfer, development, and manufacturing of TPST-2003, a dual-targeting CD19/BCMA CAR-T therapy for R/R multiple myeloma. The collaboration aims to prepare for a planned U.S. registrational study, with most development activities expected to be completed by Q3 2026 to enable a potential IND filing in Q4 2026. The partnership follows positive interim results from the ongoing P1/2a REDEEM-1 study, where all six efficacy-evaluable patients achieved a complete response, and an ORR of 100% was observed among evaluable patients across studies. Tempest plans to present results from REDEEM‑1 along with updated data from the IIT at a scientific meeting in 2026.
Source: Global Newswire PR Mar 11, 2026
March 15, 2026
Samsung Bioepis and Epis NexLab Ink Deal with G2GBIO to Develop Long-acting Drug Platform
Samsung Bioepis and its sister company Epis NexLab have entered into a research collaboration and exclusive licensing agreement with G2GBIO to develop new therapies leveraging G2GBIO’s proprietary microsphere drug-delivery technology. Samsung Bioepis will receive full licensing rights to a novel long-acting semaglutide and will have the option to license one additional asset in the future. The company will also hold first negotiation rights for three other novel assets yet to be identified. Epis NexLab will collaborate on the co-development of the long-acting microsphere platform based on G2GBIO’s technology. Financial details of the agreement have not been disclosed.
Source: Businesswire PR Mar 15, 2026

March 12, 2026
Iadademstat Receives Positive Decision for Patent Application from MPO
Oryzon Genomics has received a favorable decision from the Mexican patent office (MPO) for its patent application MX/a/2021/011610, entitled “Combinations of iadademstat for cancer therapy.” Once formally granted, the patent is expected to protect at least 2040 and expand the company’s existing coverage in Australia, Europe, Japan, and Russia. These combinations are being studied mainly in small-cell lung cancer, including regimens with checkpoint inhibitors such as atezolizumab and durvalumab. Iadademstat is being evaluated with PD-L1 inhibitors in extensive-stage SCLC (ES-SCLC) in two trials.
Source: Global Newswire PR Mar 12, 2026
Thank you for your attention! For any queries, contact us at bd@octavusconsulting.com
Interested in the presentation? Download Here

Ruchi Sengar, Senior Analyst, known for her expertise in delivering high-quality, data-driven insights. She specializes in tracking industry news, monitoring upcoming conferences, providing comprehensive post-event analyses, and developing impactful thought leadership content that enhances visibility and engagement within the global healthcare and pharmaceutical landscape.



