Key Outcomes:
- Strong late-stage clinical momentum in diverse indications, with positive phase 3 readouts for sunvozertinib in 1L EGFR exon20ins NSCLC, amlitelimab in moderate‑to‑severe atopic dermatitis, and IBI302 in nAMD, alongside phase 2 success for daridorexant in pediatric insomnia
- Pipeline progression and lifecycle expansion include Innovent advancing tigulixostat into a head‑to‑head phase 3 vs febuxostat in gout, and Ono filing ripretinib for advanced GIST in Japan based on INVICTUS data
- Regulatory milestones highlight continued focus on high‑need populations, with FDA Breakthrough Therapy designation for venglustat in type 3 Gaucher disease and Japan approval of Dupixent as the first targeted therapy for bullous pemphigoid
- Market access and formulation advances are evident with CHMP backing self‑administration of SC daratumumab in Europe and Lupin’s tentative US FDA approval for generic pitolisant tablets
- Portfolio risk‑benefit optimization is ongoing, illustrated by Eisai’s discontinuation of Tazverik in Japan due to secondary hematologic malignancy risk
- BD and strategic collaborations remain active, with Monte Rosa–J&J (MRT‑2359 + apalutamide in mCRPC), Alebund–R1 for AP306 in CKD‑related hyperphosphatemia, Novartis’ $2B+ acquisition of Pikavation’s pan‑mutant PI3Kα inhibitor SNV4818, and Insilico–Lilly’s AI‑drug discovery alliance worth up to $2.75B plus royalties

March 21, 2026
Dizal Reports Positive P3 WU-KONG28 Results for Sunvozertinib in 1L EGFR Exon20ins NSCLC
Dizal Pharmaceuticals reported positive topline findings from the P3 WU-KONG28 study evaluating sunvozertinib (Zegfrovy) as a 1L treatment for patients with NSCLC harboring EGFR exon 20 insertion mutations. The study met its primary endpoint, with sunvozertinib demonstrating a significant improvement in PFS vs. standard platinum-based chemotherapy. As a global, randomized study, these results highlight the potential of sunvozertinib as a more effective, convenient, and chemotherapy-free treatment option for this difficult-to-treat population. Detailed results are expected to be presented at an upcoming international conference, and Dizal plans to initiate discussions with regulatory authorities regarding potential NDAs.
Source: PR Newswire PR Mar 21, 2026
March 22, 2026
Innovent Advances Tigulixostat into P3 with Head-to-Head Febuxostat Study in Gout
Innovent Biologics announced that the first patient has been dosed in a P3 study of tigulixostat (IBI128, xanthine oxidase inhibitor) for gout in China. The randomized, double-blind, multi-center study will enroll 600 patients and compare tigulixostat (100 mg) directly with febuxostat (40 mg), with the primary endpoint being the proportion of patients achieving target serum uric acid levels below 360 μmol/L at 24 wks. The program builds on P2 results, where tigulixostat demonstrated superior urate-lowering efficacy and a favorable safety profile, and the data have already been published at the APLAR 2025.
Source: PR Newswire PR Mar 22, 2026
March 23, 2026
Innovent’s IBI302 Meets P3 Endpoint in nAMD
Innovent released data from P3 STAR study of efdamrofusp alfa injection in Chinese patients with neovascular age-related macular degeneration (nAMD). The study met its primary endpoint at 52 wks, showing vision improvement vs. aflibercept, while offering a key advantage of extended dosing intervals; around 73% of patients achieved 16-wk dosing, and nearly 60% showed potential for 20-wk intervals, significantly reducing treatment burden. The therapy also demonstrated comparable anatomical outcomes, a favorable safety profile, and a potential reduction in macular atrophy risk vs. aflibercept. These results position IBI302 as a promising long-acting, dual-target (VEGF and complement) treatment, with Innovent planning to move toward regulatory submission while continuing long-term follow-up.
Source: Innovent PR Mar 23, 2026
March 28, 2026
Sanofi Reports Late-Breaking P3 Amlitelimab Data at the AAD’26
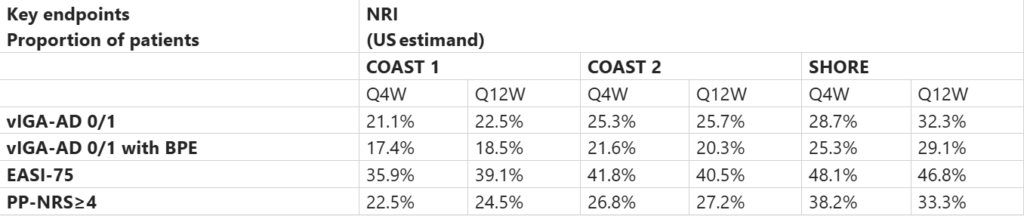
Sanofi reported new results from the P3 COAST 1, COAST 2, and SHORE studies evaluating amlitelimab in patients with moderate-to-severe atopic dermatitis (age >12 yrs). The trials showed improved skin clearance and reduced disease severity vs. placebo when amlitelimab was used alone or with topical therapies. The results were presented at the AAD’26. Primary and key secondary endpoints were evaluated at wk 24 in patients receiving amlitelimab Q4W or Q12W, with or without topical therapy. The primary endpoint was the proportion of patients achieving vIGA-AD 0/1 with ≥2-point improvement from baseline.
Primary and Key Secondary Endpoints:
Safety Profile:
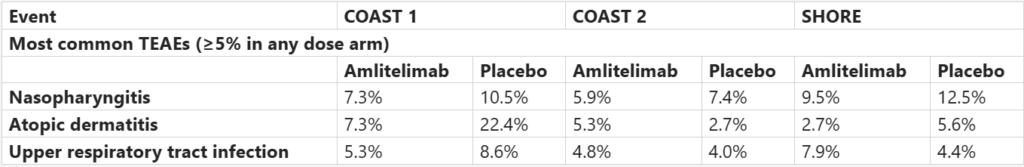
Across the three studies, pyrexia, chills, and headaches were infrequent and mostly not injection-related. Malignancy rates were <1% and similar to placebo, with no severe injection-site reactions, serious GI ulceration, or Kaposi’s sarcoma reported.
Additional Information:
- Apart from this, Sanofi reported two cases of Kaposi’s sarcoma among 3,778 amlitelimab-treated patients were identified in the P2 ATLANTIS study and P3 ESTUARY study. Treatment was discontinued in both patients, who are now recovering.
- Results from P3 ESTUARY extension study assessing Q12W maintenance dosing and long-term safety are expected in H2 2026.
- Amlitelimab remains under clinical development and has not yet been evaluated by any regulatory authority for safety or efficacy.
Source: Sanofi PR Mar 28, 2026
March 30, 2026
Idorsia Reports Positive Top-line Result of Daridorexant
Idorsia reported positive results from a P2 study of daridorexant, a dual orexin receptor antagonist, in pediatric patients (10-17 years) with insomnia disorder. Involving 165 participants, the randomized trial demonstrated a significant, dose-dependent increase in total sleep time and other sleep parameters after treatment with daridorexant (10 mg, 25 mg, 50 mg) vs. placebo. The drug was well tolerated, even at the adult dose of 50 mg, with a safety profile akin to placebo and no indications of abuse or withdrawal. Daridorexant, marketed as QUVIVIQ for insomnia, is available for adult patients in the US, Canada, and various European countries, as well as in Japan, Hong Kong, and China through partnerships. Idorsia has licensing agreements with EMS in Latin America, CTS in Israel, and Pharmalink in the Gulf region.
Source: Idorsia PR Mar 30, 2026

March 18, 2026
U.S. FDA Grants BTD to Venglustat for Type 3 Gaucher Disease
Sanofi announced that its investigational oral therapy venglustat has received Breakthrough Therapy designation from the U.S. FDA for the treatment of type 3 Gaucher disease (GD3), a rare condition with significant unmet neurological needs based on P3 LEAP2MONO study. The therapy had previously received FTD and ODD across the US, EU, and Japan. Sanofi is now preparing for global regulatory submissions in 2026.
The result of this study has already been posted on our website. (Feb 02, 2026)
Source: Sanofi PR Mar 18, 2026
March 19, 2026
Eisai Discontinues Tazverik (200 mg tablets) in Japan Over Safety Concerns
Eisai discontinued its anticancer drug Tazverik (200 mg tablets) in Japan, following safety concerns identified through global clinical and postmarketing data. The review found multiple cases of secondary hematologic malignancies associated with the drug, both in combination and as a monotherapy, including evidence from overseas trials such as P3 SYMPHONY-1 study. In line with prior voluntary withdrawals in other countries. Eisai has decided to prioritize patient safety by advising HCPs in Japan to stop current treatments and avoid initiating new ones. The company will discontinue sales once patient use ceases and will continue to guide to ensure a smooth transition for HCPs and patients.
Source: Eisai PR Mar 19, 2026
March 24, 2026
Japan Approves Dupixent for Moderate-to-Severe Bullous Pemphigoid
Regeneron and Sanofi received approval from MHLW (Japan) for Dupixent (dupilumab) for the treatment of adults with moderate-to-severe bullous pemphigoid (BP). The approval is based on results from the P2/3 LIBERTY-BP-ADEPT study, in which Dupixent demonstrated a significantly higher rate of sustained disease remission vs. placebo (18% vs. 4%; p=0.0250) at Wk 36. Adverse events were reported in 26% of patients treated with Dupixent, with conjunctivitis being the most common. Dupixent is already approved in Japan for other indications, including atopic dermatitis and asthma.
Source: Global Newswire PR Mar 24, 2026
March 25, 2026
Pitolisant Receives Tentative Approval from U.S. FDA
Lupin has received tentative approval from the U.S. FDA for its Abbreviated New Drug Application (ANDA) for pitolisant tablets, available in two dosages: 4.45 mg and 17.8 mg. The product has been confirmed to be bioequivalent to the branded drug Wakix, which is used to treat conditions like narcolepsy and excessive daytime sleepiness.
Source: Lupin PR Mar 25, 2026
March 26, 2026
Ono Files Ripretinib for GIST Approval in Japan
Ono Pharma has submitted an application in Japan for the manufacturing and marketing approval of ripretinib (DCC-2618), developed by Decipher for the treatment of advanced gastrointestinal stromal tumor (GIST) in patients whose disease has progressed after prior therapies. The application is based on results from the global P3 INVICTUS study, where ripretinib significantly improved PFS compared to placebo. Ripretinib is already approved in over 40 countries, including the U.S. and Europe.
Source: Ono Pharmaceutical PR Mar 26, 2026
March 27, 2026
CHMP Approved DARZALEX (daratumumab) SC formulation
J&J got approval from CHMP for DARZALEX (daratumumab) subcutaneous formulation as first oncology injectable in Europe for self-administration by patients or caregivers. The update to the drug’s label allows eligible patients with multiple myeloma to administer the treatment from the fifth dose onward, following proper training and approval by HCPs. The approval applies to all previously approved indications of SC daratumumab and aims to provide greater flexibility, reduce hospital visits, and ease pressure on healthcare systems while maintaining the therapy’s established safety and efficacy profile.
Source: J&J PR Mar 27, 2026

March 16, 2026
Monte Rosa Expands Oncology Pipeline with J&J Collaboration for P2 Prostate Cancer Trial
Monte Rosa Therapeutics announced a clinical supply agreement with Johnson & Johnson to support a planned P2 trial evaluating its investigational molecular glue degrader MRT-2359 + ERLEADA (apalutamide) for patients with metastatic castration-resistant prostate cancer (mCRPC), particularly those with androgen receptor mutations. Under the agreement, Monte Rosa will sponsor and conduct the trial, while Johnson & Johnson will supply apalutamide for use in the study. The trial is expected to begin in the third quarter of 2026. It aims to assess the safety and efficacy of the combination therapy, further advancing MRT-2359 as part of Monte Rosa’s oncology pipeline.
Source: Monte Rosa Therapeutics PR Mar 16, 2026
March 17, 2026
R1 Therapeutics Secures Ex-China License for Alebund’s AP306
Alebund Pharmaceuticals announced a collaboration and licensing agreement with R1 Therapeutics to advance its investigational candidate AP306 for hyperphosphatemia in chronic kidney disease (CKD), covering global markets outside Greater China. R1 receives exclusive rights to develop, manufacture, and commercialize the therapy in these regions, with financial terms including milestone payments reaching low triple-digit millions and tiered royalties in the low double-digit range on future sales. Alebund also maintains a meaningful non-dilutive equity position in R1, allowing it to benefit from long-term commercial success. R1 will take the lead in funding and executing global clinical development, including a planned P2b multiregional trial in the U.S. and China, while Alebund continues to support it as a collaborative development partner to accelerate AP306’s advancement.
Source: PR Newswire PR Mar 17, 2026
March 20, 2026
Novartis Expands Precision Oncology with Pikavation Therapeutics Deal
Novartis has agreed to acquire SNV4818, a next-generation pan-mutant-selective PI3Kα inhibitor, from Pikavation (a subsidiary of Synnovation Therapeutics) to strengthen its breast cancer pipeline. The oral drug, currently in P1/2 trials, targets mutated PI3Kα commonly found in HR+/HER2⁻ breast cancer, aiming to improve tolerability by sparing normal cells, addressing limitations of existing therapies. Designed for use alongside endocrine therapies and CDK inhibitors, SNV4818 could enable more effective and durable treatment combinations. Novartis will pay $2B upfront and up to $1B in milestones to acquire Pikavation Therapeutics, with the transaction expected to close in the 1H2026 pending regulatory approvals.
Source: Novartis PR Mar 20, 2026
March 29, 2026
Insilico Medicine and Eli Lilly Partner to Develop AI-Discovered Oral Therapeutics
Insilico Medicine and Eli Lilly entered into a licensing collaboration to develop AI-discovered oral therapeutics. Eli Lilly receives exclusive worldwide rights to develop, manufacture, and commercialize selected preclinical drug candidates discovered using Insilico’s Pharma.AI platform. The deal includes $115M upfront for Insilico, with potential development, regulatory, and commercial milestone payments that could reach up to $2.75B, plus tiered royalties on future sales. Both companies will also collaborate on multiple R&D programs, combining Insilico’s AI-driven drug discovery capabilities with Lilly’s clinical development expertise to accelerate the discovery of new medicines.
Source: PR Newswire PR Mar 29, 2026
March 31, 2026
Eli Lilly Expands Neuroscience Portfolio with Centessa Acquisition
Eli Lilly and Company has entered into a definitive agreement to acquire Centessa Pharmaceuticals to strengthen its neuroscience pipeline and expand its presence in the growing market for sleep-disorder treatments. Centessa focuses on developing orexin receptor-2 (OX2R) agonists, an emerging class of therapies designed to regulate the sleep-wake cycle and treat conditions such as Narcolepsy and Idiopathic Hypersomnia. Its lead candidate, cleminorexton (ORX750), is currently in mid-stage clinical trials and has shown promising results in improving wakefulness in patients with sleep disorders. The transaction is valued at $6.3 billion upfront, with up to $1.5 billion in additional milestone payments. Through the acquisition, Lilly aims to use its global research, development, and commercialization capabilities to accelerate the development of orexin-based therapies. The cash offer represents a 40.5% premium to Centessa’s 30-day average share price as of March 30, 2026, and has been approved by the boards of both companies.
Source: Eli Lilly PR Mar 31, 2026
Thank you for your attention! For any queries, contact us at bd@octavusconsulting.com
Interested in the presentation? Download Here

Ruchi Sengar, Senior Analyst, known for her expertise in delivering high-quality, data-driven insights. She specializes in tracking industry news, monitoring upcoming conferences, providing comprehensive post-event analyses, and developing impactful thought leadership content that enhances visibility and engagement within the global healthcare and pharmaceutical landscape.



